|
||||||||||||||||||||
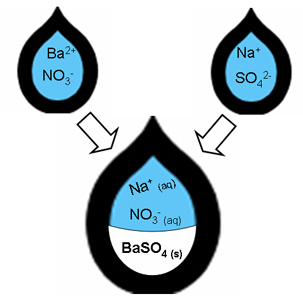
For certain combinations of solutions, a
precipitate is formed. For instance, the barium nitrate solution contains
dissolved ("aq") barium and nitrate ions, and the sodium sulfate has sodium and
sulfate ions in it. When they combine, however, the barium and sulfate ions will
unite to make a white solid ("s"), called a precipitate .