|
||||||||||||||||||||
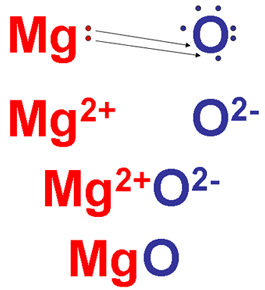
Magnesium is able to give up its two valence electrons as it reacts with oxygen. Because oxygen needs two electrons to complete its outer shell, it will accept these two electrons. The resulting compound, therefore, has a 1:1 ratio of magnesium and oxygen ions.