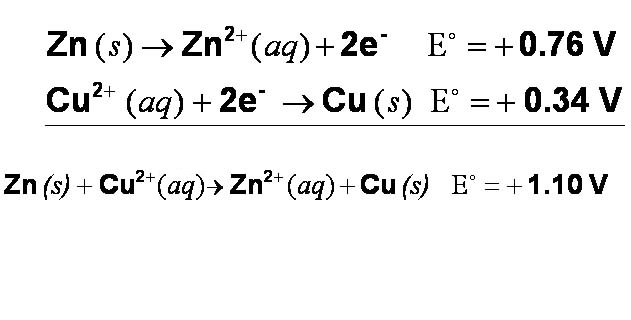|
||||||||||||||||||||
A more systematic way of describing metal reactivities in solution is to use electrochemical cell potentials. If two half-cell reactions can be added to give an overall voltage that is positive, then the combined reaction is spontaneous (i.e., it will occur without requiring a net input of energy).